Zimmer Biomet CPT®
Hip System
If you or a loved one received a Zimmer Biomet CPT® Hip System implant, you may face a significantly higher risk of serious injury, including painful thigh bone fractures. Recent warnings from the FDA and medical researchers have raised major concerns about the safety of this device, and patients harmed by the CPT Hip System may now be eligible for compensation.
At Canyon Ridge Legal, we are standing up for patients nationwide who have been impacted by defective hip implants.
What Is the CPT Hip System Recall About?
In July 2024, Zimmer Biomet issued a recall related to the instructions for its CPT Hip System Femoral Stem. New research revealed that patients implanted with this device are more than twice as likely to suffer a thigh bone fracture compared to patients with other hip implants.
Despite announcing plans to discontinue the device by December 2024, Zimmer Biomet has continued to allow the CPT Hip System to be used in surgeries — putting more patients at risk.
In response, the FDA issued a public safety alert in September 2024, warning surgeons and patients about the dangers associated with this device.
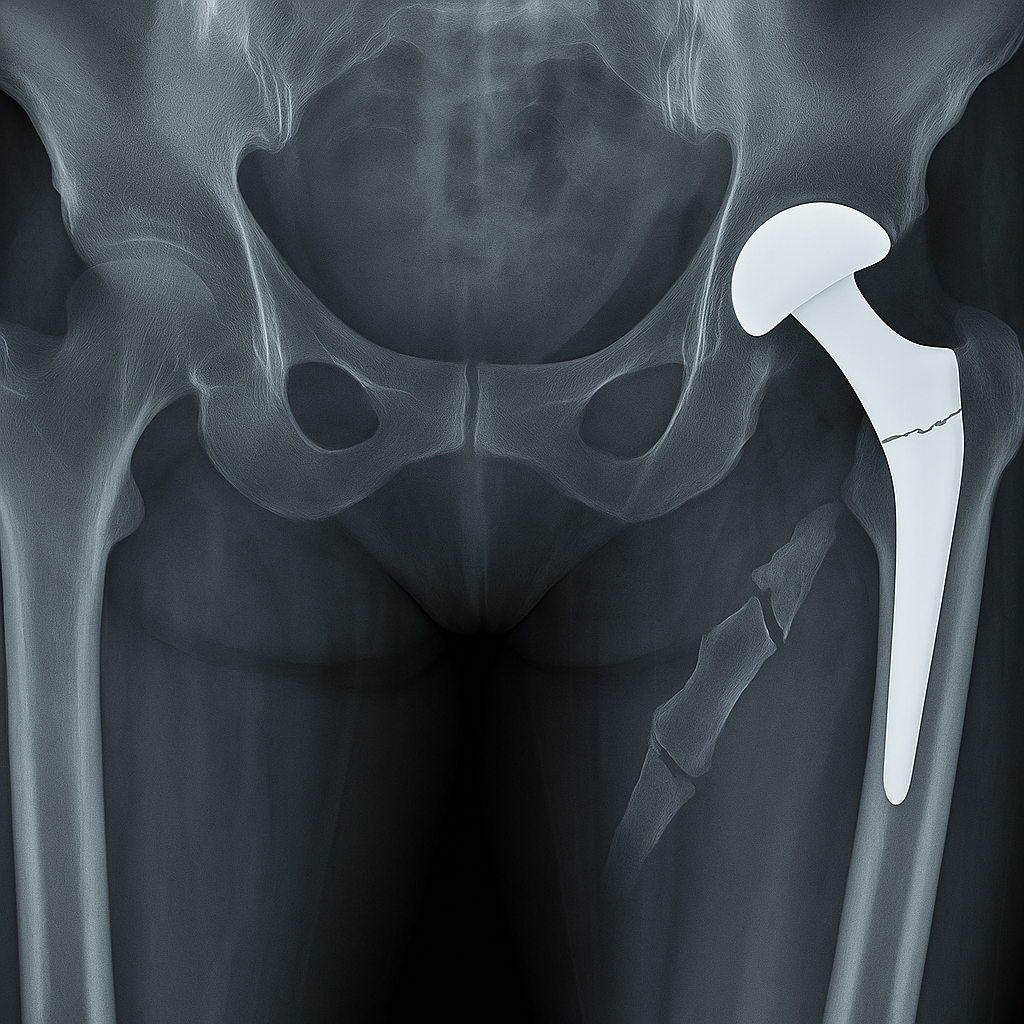
Serious Complications Linked to the CPT Hip System
Patients with a Zimmer Biomet CPT Hip implant may experience:
- Sudden or unexpected thigh pain
- Difficulty walking
- Thigh bone fractures near the implant site
- Need for revision surgery to repair the damage
- Extended recovery times and additional medical expenses
- Difficulty standing

What the FDA Recommends:
The FDA has not recommended removal of CPT Hip implants that are functioning normally.
However, patients should:
1. Monitor for new symptoms such as pain, limping, or difficulty walking.
2. Seek immediate medical attention if symptoms of device failure appears.
3. Discuss alternative implant options with their surgeons if undergoing hip replacement surgery.
Healthcare providers are advised to reserve the CPT Hip System only for cases where the benefits outweigh the known risks.

Get the Help You Deserve!
You trusted that your hip replacement would improve your quality of life.
When companies fail to deliver safe products, they must be held responsible.
Contact Canyon Ridge Legal today for a free case evaluation.
There are no fees unless we recover for you.
