IVC Filter Litigation
Canyon Ridge Legal stands with individuals nationwide who have been seriously injured by defective IVC filters. If you or a loved one has experienced complications after receiving an IVC filter implant, you may be eligible to pursue a legal claim and seek substantial compensation from the device manufacturer.
As of September 1, 2020, Canyon Ridge Legal is no longer accepting cases for defective IVC Filters
*** The information on this page is intended for educational purposes only. ***
What you need to know about the IVC filter lawsuit
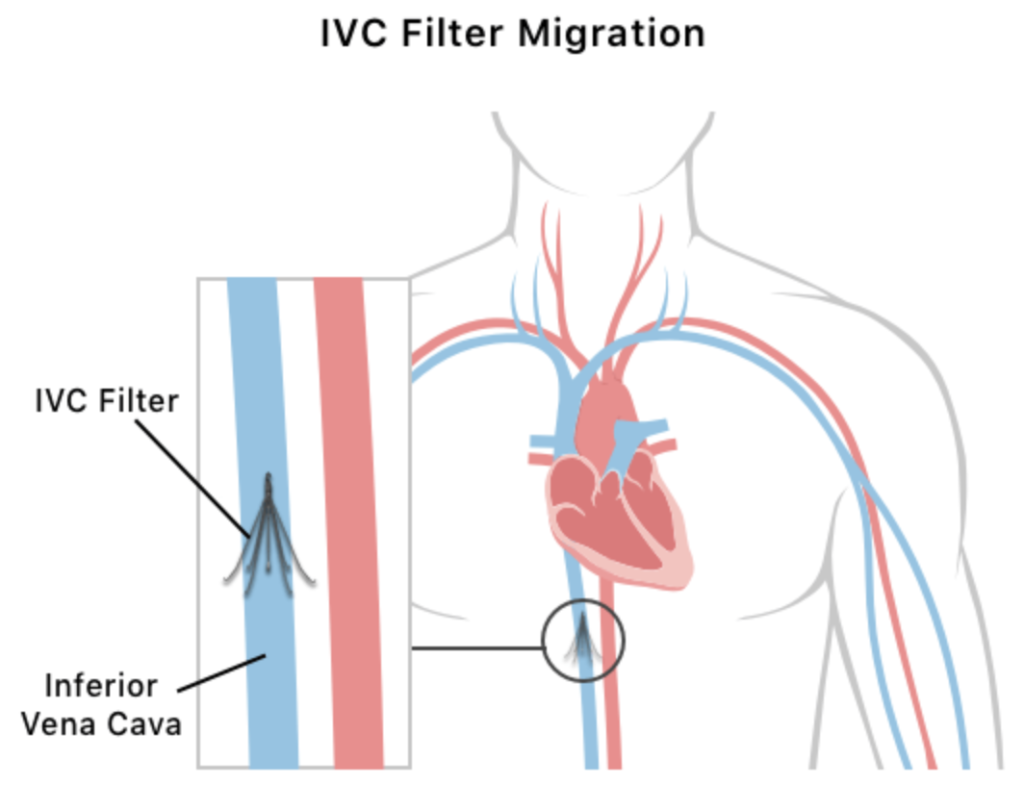
IVC (Inferior Vena Cava) filters are small, cage-like devices implanted into the body’s largest vein to help prevent blood clots from reaching the heart or lungs. Typically inserted through a vein in the groin or neck, these filters are designed to trap clots before they cause life-threatening complications.
However, serious concerns have emerged about the safety of certain IVC filters. Due to alleged design defects, some filters can migrate, tilt, or puncture the vena cava, leading to dangerous internal injuries.
Lawsuits have been filed against major manufacturers, including Cook Medical and C.R. Bard, claiming that these companies knew about the risks but failed to properly warn doctors and patients.
If you or a loved one suffered complications from an IVC filter, you may be entitled to seek justice and compensation.
Common injuries and complications
related to defective IVC filters include:
- Filter migration
- Organ / IVC perforation
- Fracturing of the IVC filter
- Tilting of the IVC filter
- Irretrievable IVC filter
- Wrongful death
- Pulmonary embolism
- IVC filter embolization
- Damage to organs
- IVC filter failure
- Deep vein thrombosis (DVT)
- Blood clots
Lawsuit Information
FDA warnings and named IVC filter manufacturers
In 2013 the prestigious Journal of the American Medical Association released a study questioning the effectiveness of IVC filters and reporting the unexpectedly high rate of failure for IVC filters. Also in 2013, the FDA issued an updated warning recommending that doctors remove retrievable IVC filters as soon as possible after the risk of pulmonary embolism has subsided. Unfortunately for some patients estimates indicate that between 10 and 20% of IVC filters cannot be safely removed.
Manufacturer
IVC Filter Model
ALN
Optional
Argon
Option Elite
Bard
Recovery
G2 | G2 Express (G2X)
Eclipse
Denali
Meridian
Boston Scientific
Greenfield
Cook Medical
Celect
Gunter Tulip
Boston Scientific
TrapEase
OptEase
What Should You Do Next?
If you are experiencing any of these complications, it is important to contact your doctor right away to determine if further treatment or revision surgery is needed. If your injuries are linked to a defective IVC filter, you may also have the right to pursue a legal claim for compensation.
Speaking with an experienced IVC filter attorney can help you understand your options and hold the manufacturer accountable. Here are a few important steps you can take to prepare for filing your claim.
- Call facility for medical records
- Document the level of your IVC filter injuries and complications
- Contact Canyon Ridge Legal for a free case evaluation
Medical Records:
If you do not already have your medical records, contact the facility where your IVC filter procedure was performed and request a full copy, including the operating report and billing statement. The operating report is especially important because it typically lists the product ID and identifies both the type of IVC filter implanted and the name of the manufacturer. This information will be critical in evaluating your legal claim.
